 A number of recent studies demonstrate a steady increase in the uptake of complementary and alternative medicines for insomnia the reasons for this increase may include dissatisfaction or concern for side effects with pharmacological treatment, previous positive experiences, and self-perceived effectiveness of alternative medicine. Recent surveys suggest that almost two-thirds of individuals with sleep problems do not consult their doctor but search for treatment advice online, and herbal medicine remains a popular choice. In contrast, most herbal medicines for insomnia and anxiety offer an exceptional safety profile, sometimes with tenfold fewer adverse events than with pharmacotherapy. It should be noted that multiple approved therapies for insomnia come with a safety warning, and some hypnotics (including, for example, barbiturates) have been abandoned because of unfavorable adverse event profiles or substance abuse. Chronic insomnia affects people across all geographies, socioeconomic levels, and cultures because of this, sleeping pills are among the most frequently prescribed medicines worldwide. Sleep also contributes to a wide range of other physiological processes, e.g., metabolic and endocrine health and the strengthening of the immune system. Healthy sleep is a crucial process for optimal cognitive performance, including attention, emotional reactivity, and learning and memory. Sleep is a fundamental physiological process required to maintain physical and emotional well-being. Currently available evidence suggests that herbal extracts may exert some of their hypnotic and anxiolytic activity through interacting with GABA receptors and modulating GABAergic signaling in the brain, but their mechanism of action in the treatment of insomnia is not completely understood. Here, we perform a systematic analysis of studies assessing the mechanisms of action of various herbal medicines on different subtypes of GABA receptors in the context of sleep control. GABA receptors are the targets of many pharmacological treatments for insomnia, such as benzodiazepines. Gamma-aminobutyric acid (GABA) is an inhibitory neurotransmitter that plays a major role in controlling different vigilance states. Research into their molecular components revealed that their sedative and sleep-promoting properties rely on interactions with various neurotransmitter systems in the brain. These herbal medicines have been shown to reduce sleep latency and increase subjective and objective measures of sleep quality. 
Centuries-old herbal treatments, popular for their safety and effectiveness, include valerian, passionflower, lemon balm, lavender, and Californian poppy. The interest in complementary and alternative medicines for treating or preventing insomnia has increased recently. These changes in the properties of GABA(A) receptor binding following antipsychotic drug administration are not consistent with those observed in schizophrenia and suggest a 'reshuffling' in GABA(A) receptor subtypes over time.Sleep is an essential component of physical and emotional well-being, and lack, or disruption, of sleep due to insomnia is a highly prevalent problem. .gif) 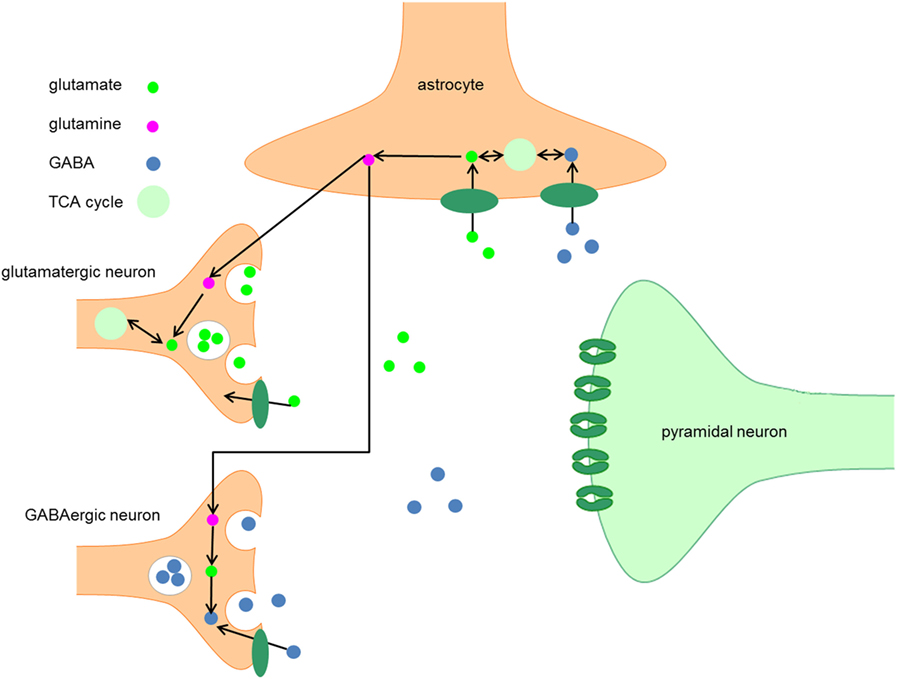
In contrast there was a delayed increase in density of benzodiazepine-sensitive GABA(A) receptors in the PFC, suggesting that antipsychotic drugs have different effects on different GABA(A) receptor subtypes. Muscimol binding was enhanced in the prefrontal cortex after 7 days but no differences were observed after longer periods of drug administration. Sections of rat brain were then labelled with muscimol, which labels the total population of GABA(A) receptors, or the benzodiazepine site ligand flunitrazepam in separate saturation binding experiments using quantitative receptor autoradiography. Male SD rats received a sucrose solution containing either haloperidol (1.5 mg/kg), olanzapine (6.5 mg/kg) or no drug daily for either 7, 14 or 28 days. As antipsychotic drugs have delayed maximal therapeutic effects we also examined different drug treatment periods. To test this, we examined the effects of administering a typical (haloperidol) and an atypical (olanzapine) antipsychotic drug on the GABA(A) receptor agonist (orthosteric) and benzodiazepine (allosteric) binding sites in rat prefrontal cortex. However, long-term antipsychotic drug use in schizophrenia may underlie these changes. Changes in GABA(A) receptors are observed in schizophrenia, with benzodiazepine-sensitive GABA(A) receptor subtypes being affected differently to other subtypes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
